
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Join Us at Booth N5M07 | November 12–14, 2025 | Chongqing International Expo Center
Tianming Pharmaceutical Group is proud to announce our participation in the 93rd China International Pharmaceutical Raw Materials, Intermediates, Packaging, and Equipment Trade Fair (API China).
The event will be held from November 12 to 14, 2025, at the Chongqing International Expo Center. We warmly invite all our global partners, customers, and industry peers to visit us at Booth N5M07.
Founded with a strong commitment to innovation and quality, Tianming Pharmaceutical Group has become a leading manufacturer of pharmaceutical intermediates and active pharmaceutical ingredients (APIs).
With years of experience in research, development, and large-scale production, we specialize in providing high-quality intermediates for therapeutic areas such as cardiovascular, anti-diabetic, and anti-tumor drugs.
Our state-of-the-art facilities operate under strict GMP standards, ensuring that every product meets global pharmaceutical quality requirements. We serve a wide range of customers across Asia, Europe, and North America, and are continuously expanding our partnerships through technology innovation and reliable supply capabilities.

API China is one of the most influential exhibitions in the pharmaceutical industry, connecting global manufacturers, researchers, and buyers. Visiting Tianming’s booth provides an excellent opportunity to:
We believe face-to-face communication is the foundation of lasting partnerships, and we look forward to sharing how Tianming’s professional expertise can support your pharmaceutical development goals.
Email: sunqian0123@gmail.com
WhatsApp: +86 176 6371 3557
We Look Forward To Your Visit!

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
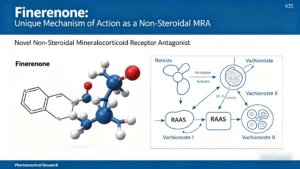
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.