
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Table of Contents

Within modern oncology drug development systems, the importance of Oncology Intermediates is being increasingly recognized by pharmaceutical companies. Many quality risks, scale-up failures, or regulatory hurdles do not originate at the API or formulation stage, but are often “set in motion” much earlier during the intermediate manufacturing phase.
Based on extensive production experience serving oncology clients, oncology intermediates are not merely “scaled-up versions of standard intermediates.” They demand significantly higher levels of process understanding, quality control, compliance capabilities, and accumulated expertise.
This article systematically outlines the core technical and quality challenges facing oncology intermediates from a manufacturer’s perspective, integrating industry consensus with real-world project experience.
The molecular characteristics of oncology drugs inherently confer greater complexity to their upstream intermediates. According to publicly available drug development statistics, a significant proportion of small-molecule anticancer drugs currently in development or on the market exhibit the following features:
This implies that if route selection is inappropriate or control fails during the intermediate stage, there is virtually no “recovery margin” in the subsequent API stage. In actual projects, we have repeatedly observed downstream clients investing substantial resources in the API stage only to be forced to rework due to irreversible impurity structures in a critical intermediate.
Therefore, in oncology projects, intermediates are not cost centers but the frontline of risk control.
Oncology intermediates often involve the following high-risk reaction types:
These reactions are extremely sensitive to temperature, feed ratios, solvent purity, and reaction sequence. Reactions controllable under laboratory conditions often exhibit significantly altered byproduct ratios when scaled up to kilogram or ton-scale production.
In actual manufacturing, we have observed:
Temperature fluctuations of just 2–3°C can double the proportion of critical byproducts. Such changes cannot be fully predicted through theoretical modeling alone and require multi-batch process optimization and trend tracking.
Unlike conventional therapeutics, oncology drugs typically exhibit lower tolerance for impurities. Certain potential genotoxic impurities (GTIs), once formed and “embedded” within the molecular skeleton during the intermediate stage, become extremely difficult to completely remove through subsequent crystallization or purification steps.
This explains why an increasing number of regulated market clients now directly request the following during audits:
Industry experience indicates that 80% of impurity issues in oncology APIs can be traced back to route selection and control strategies during the intermediate stage.
Oncology intermediates often undergo rapid scale-up from gram-scale to kilogram-scale to hundred-kilogram-scale. Common issues during scale-up include:
Across multiple projects, we observed:
A process deemed “feasible” in small-scale trials does not guarantee industrial-scale stability. Truly reliable oncology intermediate processes must undergo deliberate stress testing under extreme conditions prior to scale-up—not merely “success under normal conditions.”
Increasingly, oncology drug projects establish comprehensive CMC strategies during early clinical phases. This directly elevates client requirements beyond mere “usability” to include:
This represents a global trend in oncology drug development, not isolated “high demands” from individual clients.
In oncology intermediate projects, single HPLC data is insufficient for quality assessment. Mature industry practices typically include:
These data are not merely “audit compliance measures,” but rather tools that help manufacturers identify potential risks early on, preventing uncontrollable quality deviations later.
Based on extensive project experience, we believe production systems with genuine oncology intermediate capabilities typically exhibit the following characteristics:
These capabilities are not achieved through short-term investment but are the result of long-term project accumulation.
The technical complexity and quality demands of Oncology Intermediates are increasingly diverging from conventional intermediates. This field tests not only equipment or certifications, but also the depth of understanding regarding reaction mechanisms, quality risks, and process evolution.
Drawing from fifteen years of experience in intermediate manufacturing, we unanimously believe that for oncology drug projects, selecting a partner who truly understands the value of intermediates and possesses practical expertise is often more critical than merely comparing prices.
Cooperation online: sunqian0123@gmail.com
WhatsApp: +86 176 6371 3557

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
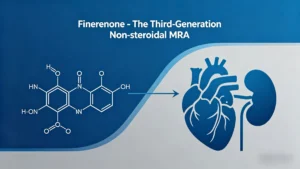
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
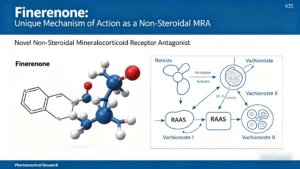
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.