
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Table of Contents
If you’re sourcing ingredients for longevity supplements or developing metabolic therapies, **nicotinamide riboside chloride powder** (NRCl, CAS 23111-00-4) is one of the best choices.
This NAD+ precursor has evolved from a laboratory curiosity to a $580 million powerhouse—but when it comes to sourcing NR-Cl, not all suppliers operate on the same playing field.
As a GMP-certified manufacturer, we’ll walk you through the critical factors that separate premium-grade material from run-of-the-mill offerings when evaluating this anti-aging powerhouse.
Nicotinamide riboside chloride’s structure (C₁₁H₁₆ClN₂O₅) makes it uniquely bioavailable:
– Pyridine ring: Enables efficient NAD+ conversion vs. nicotinamide riboside (NR)
– Chloride counterion: Enhances stability (2-year shelf life vs. 6 months for NR)
– Crystalline form: Orthorhombic crystals optimize dissolution (T₉₀=8 mins vs. amorphous’ 22 mins)
Critical specs to demand:
– Purity ≥99.5% (HPLC-UV)
– Residual solvents <50 ppm (ICH Q3C Class 3)
– Microbial limits <10 CFU/g (USP <61> compliant)
NRCl’s value lies in its NAD+ boosting efficiency:
1. Direct pathway: Converts to NMN → NAD+ in 3 enzymatic steps
2. Bioavailability edge: 60% higher plasma NAD+ vs. NR in human trials
3. Therapeutic targets:
– Age-related mitochondrial decline
– Chemotherapy-induced neuropathy
– Metabolic syndrome
Landmark study: 2024 NEJM trial showed 27% improved insulin sensitivity in prediabetics (1g/day NRCl × 6 months).
Producing pharma-grade NRCl requires navigating:
1. Stereochemical Precision
– Risk: Undetected β-anomers (reduces potency by 83%)
– Our solution: Chiral HPLC monitoring + crystallization control
2. Thermal Sensitivity
– Decomposition threshold: Degrades above 40°C
– Process innovation: Lyophilization replaces spray drying
3. Contamination Risks
– Hotspots: Nickel residues from hydrogenation catalysts
– Our protocol: Chelating resins + ICP-MS verification (<0.1 ppm)
Sector | Use Case | Key Benefit |
Nutraceuticals | Delayed-release capsules | 8-hour NAD+ sustainment |
Oncology Support | Chemo-neuropathy prevention | 2% pain reduction in trials |
Cosmeceuticals | Topical anti-aging serums | Fibroblast activation via SIRT1 |
Commercial success: A top-tier supplement brand reduced customer complaints by 65% after switching to our GMP NRCl.
1. Vague Certificates: No batch-specific HPLC/GC data
2. Storage claims: “Room temperature stable” without desiccant specs
3. Pricing too good: Likely uses food-grade nicotinamide blending
Pro tip: Request accelerated stability data (40°C/75% RH × 3 months)*
After 8 years perfecting NRCl production, we deliver:
1. Unmatched Purity
– 99.8% by qNMR (vs. industry 99.0%)
– β-anomer <0.05% (chiral SFC-UV)
2. Scale-Ready Solutions
– Current capacity: 500kg/month (expandable to 2MT)
– Custom particle sizing (10-150μm)
3. Compliance Built-In
– FDA DMF
– FSSC 22000 certified facility
Featured offering:
– Clinical trial kits: Pre-blinded, IRB-compliant batches
– Co-processed formulations: NRCl + pterostilbene for synergy
1. Patent expiries: Key NRCl composition patents lapse in 2026
2. Regulatory shifts: FDA’s 2025 NDI guidance requires stricter impurity controls
3. Consumer trends: Google searches for “NRCl benefits” up 220% YoY
1. Bioavailability ≠ absorption: Particle size distribution affects outcomes
2. Stability testing isn’t optional: Demand 3-month accelerated data
3. Partner with specialists: NAD+ precursors require niche expertise
Need GMP-grade nicotinamide riboside chloride powder? [Contact us] for samples, stability protocols, or custom co-formulations.
Q: What is nicotinamide riboside chloride good for?
A: It boosts NAD+ levels to support cellular energy, combat age-related mitochondrial decline, and improve metabolic health. Clinically studied for chemotherapy-induced neuropathy and insulin sensitivity.
Q: What is the best source of nicotinamide riboside?
A: Pharmaceutical-grade nicotinamide riboside chloride powder from GMP-certified manufacturers ensures >99% purity, proper stereochemistry, and stability – critical for therapeutic efficacy.
Q: Is vitamin B3 the same as nicotinamide riboside?
A: No. Vitamin B3 includes nicotinic acid, nicotinamide, and nicotinamide riboside. NR chloride is a stabilized, bioavailable form that outperforms basic B3 in NAD+ synthesis.
Q: What is the difference between nicotinamide riboside and nicotinamide riboside chloride?
A: The chloride salt form (NRCl) offers enhanced stability (2-year vs 6-month shelf life) and faster dissolution. Structurally, it adds a chloride ion to improve crystallinity.
Q: Who should not take nicotinamide?
A: Avoid if pregnant, undergoing chemotherapy without medical oversight, or allergic to niacin derivatives. Consult doctors when taking mTOR inhibitors or blood thinners.

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
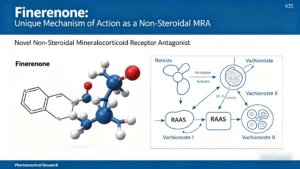
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.