
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Table of Contents
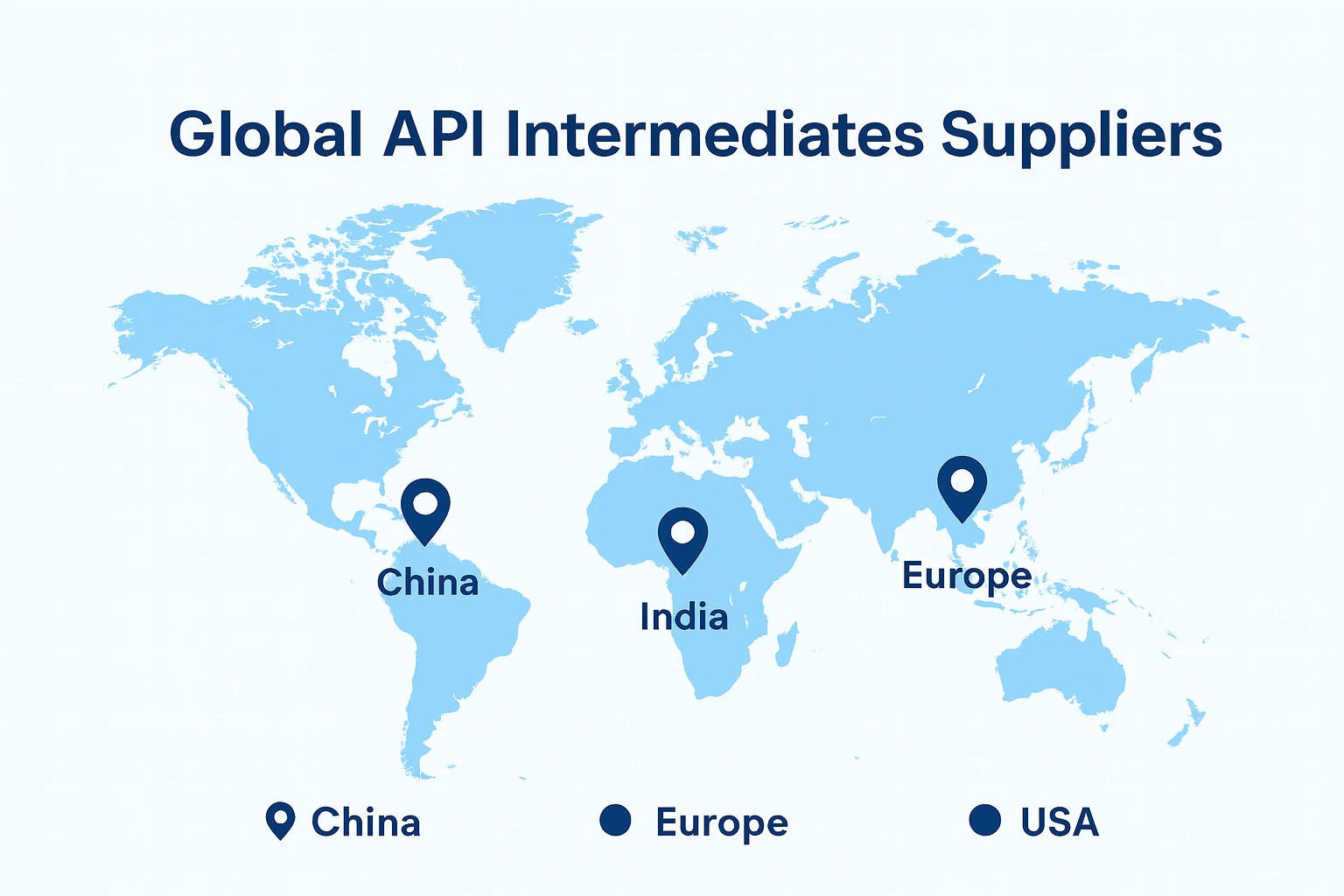
As global pharmaceutical supply chains become increasingly interconnected, API intermediates continue to play a critical role in supporting economical and compliant drug development. With regulatory frameworks tightening across major markets—and more regions shifting toward value-added API production—the demand for reliable intermediate suppliers continues to rise.
This article aims to move beyond exaggerated claims and instead provide a fact-based overview of major API intermediate suppliers worldwide, regional manufacturing strengths, and the key criteria companies use when evaluating supply chain partners.
According to multiple industry sources—including IQVIA and Evaluate Pharma reports from 2023–2024—the global API and intermediate manufacturing market has indeed shown steady, consistent growth. Several concrete factors are driving this trend:
Small molecules still account for over 70% of global prescription medicines, forming the backbone of pharmaceutical demand.
Production capacity continues to rise across India, China, Europe, and Southeast Asia.
Oncology, cardiovascular, metabolic, and CNS drug pipelines are expanding rapidly—all of which rely heavily on high-purity intermediates.
More pharmaceutical companies are outsourcing early-stage synthesis, process optimization, and scale-up to specialized partners.
Together, these industry-verified trends highlight the need for a clearer and more transparent reference of global API intermediate suppliers—one that reflects real manufacturing capabilities and regional specializations.
The global distribution of API intermediate manufacturers is not random; it reflects historical development, industrial clustering, and regulatory evolution:
With a strong chemical manufacturing foundation and major production clusters in Zhejiang, Jiangsu, and Shandong, China remains one of the world’s leading producers of pharmaceutical intermediates.
According to data from the China Chamber of Commerce for Import and Export of Medicines & Health Products (CCCMHPIE), China supplies a significant portion of global intermediates for cardiovascular, diabetes, antiviral, and oncology APIs.
As one of the world’s largest exporters of generic pharmaceuticals, India plays a vital role in the API intermediate supply chain.
Data from the Indian Pharmaceutical Alliance shows competitive strengths in intermediates for anti-infectives, oncology drugs, and chronic disease treatments.
Europe hosts a group of highly specialized manufacturers focused on high-value, highly regulated intermediates. Companies in Germany, Switzerland, and Italy emphasize purity, advanced technologies, and strict regulatory compliance.
The U.S. is home to several high-performance CDMOs and niche intermediate manufacturers. While overall production volume is smaller compared to Asia, the technological barrier and regulatory intensity are higher, especially for complex, high-potency, or controlled-substance intermediates.
(Based on Public Regulatory and Industry Data)
The following companies are representative examples of global API intermediate suppliers. Their inclusion is based on publicly available regulatory filings, DMF databases, industry reports, and corporate manufacturing disclosures.
This list reflects publicly available regulatory and industry information from 2023–2024 and is intended as a factual reference rather than a promotional ranking.
Companies procuring intermediates—whether for generic drugs or innovative molecules—generally rely on measurable standards rather than subjective impressions. The criteria below align with expectations from FDA, EMA, and ICH guidelines.
These elements form the foundation of modern API intermediate sourcing and are essential for minimizing supply chain risks.
A verified, fact-based supplier list is crucial for:
Inaccurate or unverifiable supplier information can lead to non-compliance, supply interruptions, or audit failures.
A transparent, data-supported list gives buyers confidence and provides a valuable tool for risk mitigation.
In an increasingly complex pharmaceutical manufacturing ecosystem, an accurate global API intermediates supplier reference is essential for procurement teams and R&D professionals.
By relying on public regulatory data, verified industry sources, and proven operational performance, companies can more effectively identify reliable partners and manage supply chain risks.
China, India, Europe, and the United States remain the core regions for intermediate manufacturing—each with distinct strengths.
For companies seeking compliant, high-purity intermediates—particularly in cardiovascular, diabetes, oncology, and complex synthesis projects—choosing a technically capable supplier with a strong regulatory track record is fundamental.
A transparent and data-driven evaluation process not only increases long-term project success but also strengthens confidence throughout the entire pharmaceutical supply chain.
For organizations looking for trusted and compliant intermediate solutions, Tianming Pharmaceutical welcomes the opportunity to collaborate.
Email: sunqian0123@gmail.com
WhatsApp: +8617663713557

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
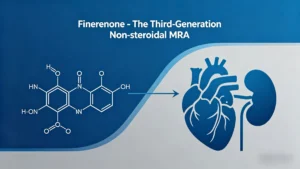
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
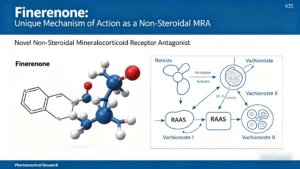
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.