
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Against the backdrop of the global rise in the incidence of diabetes and obesity, Orforglipron—an oral small-molecule partial agonist of the GLP-1 receptor—is leading a transformation in the treatment paradigm for metabolic diseases.
Traditional GLP-1 receptor agonists are primarily administered via injection, which has limitations such as low patient compliance and stringent storage conditions. Orforglipron achieves three major technical breakthroughs through its innovative small-molecule non-peptide structural design:
Weight loss efficacy
Phase II clinical trial (36 weeks) showed:
Glycemic Control
In the Phase III ACHIEVE-1 trial (40 weeks):
Shandong Tianming Pharmaceutical Group has successfully achieved full-scale commercial production of the core intermediate for the antidiabetic drug Orforglipron after a year of intensive research and development.
The R&D team improved the yield of the key segment to 92% through innovative synthesis route design, representing a 40% increase over traditional processes. They also optimized the catalytic system using continuous flow reaction technology, controlling impurity generation to below 0.05%.
The Quality Center has established 16 online monitoring points to ensure the purity of the intermediate remains above 99.7%, fully compliant with the standards of the European and American Pharmacopoeias.
Currently, annual production capacity has exceeded 15 tons, capable of meeting the production needs for 30 million doses of oral formulations globally.
This achievement not only breaks foreign technological monopolies but also reduces waste emissions by 65% through green process upgrades, setting a new benchmark for the industrialization of oral peptide-based drugs and significantly advancing the transition of diabetes treatment from injection to oral therapy.
Tianming Pharmaceutical Group has ample inventory of Orforglipron intermediate series products. We welcome inquiries and cooperation discussions from all sectors!

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
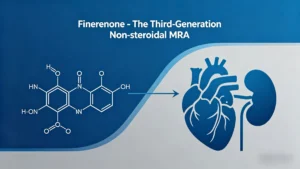
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
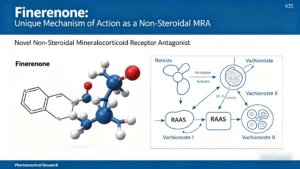
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.