Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
3-(Pyrrolidin-2-yl)pyridine;Nicotine metabolite; 3-Pyridyl-2-pyrrolidine; Nicotine Analog; Nicotinic Receptor Agonist
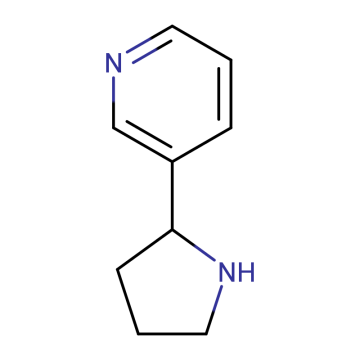
CAS:494-97-3
Chemical Formula:C9H12N2
MW:148.21
Availability: R&D; Commercialization

⬤ CAS No.494-97-3
⬤ Packaging type:
20kg barrel;5kg barrel;1kg bag;500g bag ; Customized Packaging
| Product Name | Nornicotine |
| CAS Number | 494-97-3 |
| Molecular Formula | C9H12N2 |
| Molecular Weight | 148.21 |
| Synonyms | (±)-Nornicotine, (S)-Nornicotine, 3-(Pyrrolidin-2-yl)pyridine, Nornicotine Base |
| Density | 1.043 g/cm³ |
| Boiling Point | 269.999 ºC at 760 mmHg |
| Flash Point | 111.291 ºC |
| Storage | Store in a cool, dry, well-ventilated area; separate from foodstuffs |
| Purity | ≥96.0% or ≥97.0% |
| Packaging | 10mg, 25mg, 50mg, 100mg |
| Application | Scientific research, drug development, etc. |
| Product Identification | Nornicotine |
| CAS Number: 494-97-3 | |
| Intended Use | For scientific research and development purposes only. Not for human or animal consumption. |
| Safety Precautions | Handle with caution. Wear appropriate personal protective equipment (PPE) including chemical-resistant gloves, safety glasses, and lab coat. |
| Avoid contact with skin, eyes, and mucous membranes. | |
| Work in a well-ventilated area. | |
| In case of contact, immediately flush affected area with water and seek medical attention. | |
| Storage Conditions | Store in a cool, dry, and well-ventilated area. |
| Keep away from heat, sparks, and open flames. | |
| Store separately from food, beverages, and other consumables. | |
| Handling Instructions | Use only in a dedicated laboratory setting under the supervision of a qualified professional. |
| Follow all local regulations and guidelines for handling hazardous chemicals. | |
| Use appropriate tools and equipment for weighing and transferring Nornicotine. | |
| Disposal | Dispose of Nornicotine and any contaminated materials according to local regulations and guidelines for hazardous waste disposal. |
| Do not dispose of in regular trash or down drains. |
| Product Name | Nornicotine |
| Purity | ≥96.0% or ≥97.0% (as per specifications) |
| Appearance | [White Powder] |





Exceptional Product Quality
“We have been sourcing pharmaceuticals from Tianming Pharmaceutical for several years now, and their product quality has consistently exceeded our expectations. Each batch undergoes rigorous testing, ensuring we receive only the highest quality products. This reliability has significantly contributed to our trust and satisfaction with their services. Highly recommended!”
June 15, 2023





Outstanding Technical Guidance
“The technical guidance provided by Tianming Pharmaceutical’s team has been invaluable to our research and development projects. Their experts are knowledgeable, responsive, and always willing to assist with detailed explanations and support. Their guidance has enabled us to optimize our processes and achieve remarkable results. We couldn’t ask for a better partner.”
June 15, 2023





Superior Service Experience
“Our experience with Tianming Pharmaceutical has been nothing short of excellent. Their customer service team is exceptionally responsive and dedicated to meeting our needs. From initial consultation to after-sales support, they have provided seamless, professional service. Their commitment to customer satisfaction is truly commendable.”
June 15, 2023





Precise Product Specifications
“Tianming Pharmaceutical has impressed us with their adherence to precise product specifications. Each order is meticulously packaged and delivered exactly as requested, ensuring that we receive products that perfectly meet our requirements. Their attention to detail and commitment to accuracy have made them a trusted supplier for our business.”
June 15, 2023
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.