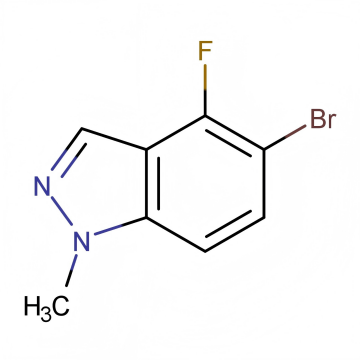
Understanding MOQ for Pharmaceutical Intermediates: What Every API Buyer Should Know Before Placing an Order
A manufacturer’s inside look at where minimum order quantities come from, what’s reasonable at each development stage, and 5 practical strategies to get better terms.