Finerenone Drug Class: What Makes This MRA Different from the Rest
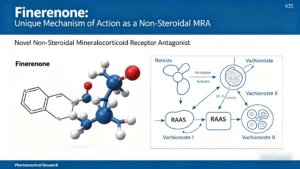
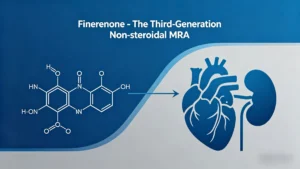
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.