
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Table of Contents
The health supplement industry has been quite lively recently, with a focus of debate garnering considerable attention. After reviewing numerous articles and videos, we’ve discovered that the core of the controversy stems from a confusion between “health foods” and “drugs.”
Today, Tianming Pharmaceuticals would like to share some basic comparisons between drugs and health foods from a professional perspective for your reference and consideration, hoping to inform your future consumer decisions.
According to the Drug Administration Law, drugs are substances used to prevent, treat, or diagnose human diseases, or to purposefully regulate physiological functions. Common examples include antibiotics, antihypertensive drugs, and antipyretics.
According to the Food Safety Law, health foods are foods that claim to have specific health benefits or are intended to supplement vitamins or minerals. They are not intended to treat diseases. Common examples include calcium supplements, probiotics, and fish oil.
Please note: National regulations do not allow ordinary foods to claim “health benefits.” In other words, health benefits must be legally declared and approved, and cannot be exaggerated or implied. A preliminary indicator of legal and compliant health foods can be found by looking at their packaging: a blue cap (“blue health food logo”) is printed on the outer packaging.
Both pharmaceuticals and health foods require registration or filing procedures before they can be marketed, but the complexity, timeframe, and investment costs differ significantly.
Drug registration
Typically, a 5-10 year R&D cycle, including clinical trials, quality control, safety assessments, and the preparation of extensive technical documentation, is required before a registration application can be submitted. The overall investment can often reach hundreds of millions or even billions of yuan, resulting in a long process and high barriers to entry.
Health Food Registration/Filing
Comparatively speaking, the registration or filing process for health foods is more streamlined. It generally takes about two to three years and focuses primarily on functional evaluation, safety, and product stability. The investment costs range from several million to tens of millions of yuan, making it more manageable than for pharmaceuticals.
Take artemisinin-based drugs as an example. Before they can be marketed, they must complete a rigorous research and evaluation process, including:
In short, for a drug to be approved for marketing, its effectiveness and safety must be fully validated, and its risks must be within manageable limits.
Health foods, on the other hand, do not have these requirements and typically do not undergo human clinical trials. Their efficacy is often supported by animal studies, functional evaluation tests, or existing literature, and unlike drugs, their effectiveness in humans cannot be directly proven.
The figure below compares the R&D investments for drugs and health foods. The high R&D costs of pharmaceutical companies, coupled with a failure rate exceeding 90%, justify the high prices of originator drugs in this context.
Dimensions | Drugs | Health Foods |
Fund Size | Huge investment | Relatively low |
Risk Level | The failure rate exceeds 90%, and failure is common in the clinical stage | If the raw materials are safe, the success rate will be higher |
Cost | Clinical trials account for more than 60% of total costs | Functional evaluation tests account for the majority of costs |
Drugs have a clear therapeutic purpose and treat a range of diseases. Health foods’ core purpose is to maintain and improve health. The packaging clearly states that health foods are not drugs and cannot replace drugs in treating diseases.
The saying “all medicines are toxic” is true for most drugs. Drugs are used to treat diseases and are generally potent and effective, but they may also have certain side effects. Therefore, it is recommended to “avoid medication if you are not ill, but seek medical attention promptly if you are ill.”
One of the core evaluation factors for health foods is safety. They are intended to help improve sub-health and regulate body functions, not to treat diseases.
If you only have minor or temporary discomfort, it is recommended to try compliant health foods as a part of your daily health management.
Important Notes:
1. Health foods are prohibited from making claims about their therapeutic effects. Any promotion of “treating XX disease” is illegal (commonly seen in false advertising).
2. Regarding “food-drug” Chinese medicines, please note: when used as medicines, they are regulated as such (e.g., Panax notoginseng powder is used to stop bleeding). When used as food, only ingredients listed in the catalogue may be used, and no functional claims may be made.
3. Consumer rights protection: Drug quality issues are governed by the Drug Administration Law, while health food disputes are governed by the Food Safety Law.
Welcome to Tianming Pharmaceutical Group for more industry knowledge!

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
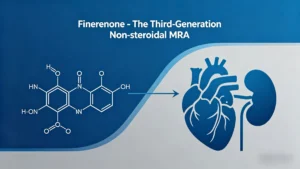
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
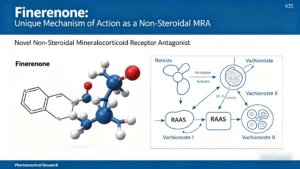
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.