
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
In the field of API and intermediate production, Continuous Flow Manufacturing (CFM) is revolutionizing traditional production models with its disruptive advantages.
This technology utilizes equipment such as microreactors and continuous stirred tank reactors to transform batch production into dynamic continuous processes, significantly enhancing efficiency and safety while serving as a critical stepping stone for the pharmaceutical industry’s transition toward intelligent manufacturing.
Continuous flow systems achieve millimeter-level reaction control by real-time monitoring of parameters such as temperature, pressure, and pH.
For example, in nitration reactions, traditional batch processes are prone to safety incidents due to heat accumulation, while microchannel reactors can reduce reaction time to seconds, improve heat diffusion efficiency by 90%, and maintain product purity above 99.5%.
Equipment footprint reduced by 70%: After renovation, the production capacity of a cephalosporin intermediate production line increased from 50 kg/day to 200 kg/day per unit.
Solvent consumption reduced by 40%: Continuous extraction processes replaced multi-stage liquid-liquid separation operations.
Labor costs reduced by 65%: Automated systems enable 24/7 unmanned operation.
Continuous flow processes enable VOC emissions to comply with EU REACH regulations. A certain anticancer drug intermediate production line achieves zero wastewater discharge through a closed-loop water circulation system.
Reaction type Traditional process defects Continuous flow solutions
High-risk reactions Risk of heat accumulation Microchannel enhanced heat transfer (>10^4 W/m²K)
Multi-step sequential reactions Intermediate separation losses Integrated online purification modules
Photochemical synthesis Low light source utilization Microfluidic photoreactor (efficiency improved by 8 times)
Flow rate range: 0.1–1000 kg/day (must match production line scale)
Material compatibility: Hastelloy C-276 for highly corrosive systems
Module scalability: Supports parallel capacity expansion
A company adopted a continuous flow process to produce a key intermediate for remdesivir, achieving:
Reduction of reaction steps from 7 to 3
Total yield increased from 52% to 83%
Production cycle compressed from 14 days to 72 hours
Through continuous crystallization technology, a steroid drug production line achieved:
Crystal form control precision of ±2 μm
Filter efficiency improved by 300%
Batch-to-batch variation coefficient (RSD) <1.5%
Scale effects: Differences in fluid behavior within microchannels (recommended to validate via cold-mold experiments)
Residence time distribution: Optimized pipeline design using PFR models
Near-infrared spectroscopy for online monitoring
Raman spectroscopy for real-time analysis of reaction progress
Machine learning algorithms for predicting product quality
Digital twin technology for virtual commissioning
5G+ industrial internet enabling remote control
Blockchain technology ensuring traceability of production data
Electrochemical continuous synthesis technology (e.g., CO₂ resource utilization)
Biocatalysis and continuous flow coupling systems
Solar-driven photochemical reaction devices
As a technical service provider with over a decade of experience in the API manufacturing sector, we have successfully implemented 23 continuous flow retrofit projects, helping clients achieve:
An average investment payback period of 18 months
100% FDA/EMA certification pass rate
A 55% reduction in carbon emissions intensity
If you are seeking to innovate your production model, please contact our technical team to obtain customized continuous flow solutions. Let us work together to drive the pharmaceutical industry toward greater efficiency, safety, and sustainability.

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
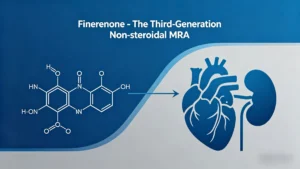
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
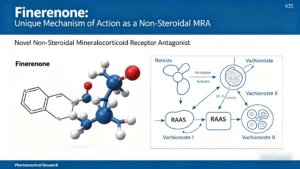
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.