If you’ve been following the GLP-1 space at all, you’ve probably seen these two names pop up everywhere: tirzepatide (the injectable that’s already on the market) and orforglipron (the oral pill still in late-stage trials). People keep asking which one is better. But here’s the thing – “better” depends entirely on what you’re measuring.
I’ve spent years working on the chemistry behind GLP-1 compounds at Tianming Pharmaceutical. When I first saw orforglipron’s structure, I wasn’t just impressed by the weight loss numbers.
What really got my attention was that someone finally cracked the code on an oral, small-molecule GLP-1 agonist that could actually compete with injections. That’s not a small achievement.
So let’s break down how these two actually compare – not just the headlines, but the real data behind them.
How They Work: One Pathway vs Two
Tirzepatide is a dual agonist. That means it hits two receptors at once – GLP-1 and GIP. According to a 2024 systematic review on its pharmacology, activating both pathways explains why tirzepatide often leads to better blood sugar control and more weight loss compared to drugs that only target GLP-1.
Orforglipron, on the other hand, only targets GLP-1. But here’s what makes it different from every other GLP-1 drug out there: it’s a small molecule. A paper in Science Translational Medicine showed that orforglipron binds to the human GLP-1 receptor with a Ki of just 1 nM – that’s very strong binding.
The same study noted it has low intrinsic efficacy for effector activation and barely recruits β-arrestin. In plain English? That might mean less receptor desensitization over time.
The Numbers You Actually Care About
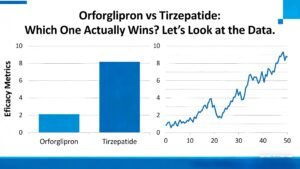
Let me just put the key trial results side by side so you can see the difference clearly.
Metric | Orforglipron (36 mg, daily pill) | Tirzepatide (15 mg, weekly injection) |
Main trial | ATTAIN-1 (n=3,127, 72 weeks) | SURMOUNT-1 (n~2,539, 72 weeks) |
Average weight loss | -12.4% | -20.9% |
Placebo-adjusted loss | -9.5 to -11.5 percentage points | -17.8 percentage points |
Lost at least 10% | 55–60% | ~75% |
Lost at least 20% | ~18% | ~57% |
GI side effects | Common (nausea, vomiting, diarrhea) | Common (mostly mild to moderate) |
Stopped due to side effects | ~10% (mostly GI) | 6–7% at highest dose |
How you take it | Daily pill, no food/water restrictions | Weekly shot, needs training |
Where these numbers came from:
The orforglipron data is from Lilly’s August 2025 press release on the ATTAIN-1 trial. The tirzepatide numbers are from Lilly’s published SURMOUNT-1 data. A head-to-head comparison in Cardiovascular Diabetology – Endocrinology Reports also compiled these directly from both trials.
What Indirect Comparisons Tell Us (And What They Don’t)
A network meta-analysis published in Circulation looked at 25 RCTs with nearly 16,000 people. It compared 11 different weight loss drugs. At 1 to 1.5 years, tirzepatide had a SUCRA score of 0.86 – meaning it had a very high chance of being among the best. Orforglipron scored 0.47. Tirzepatide also beat weekly semaglutide by 3.2% and liraglutide by a huge 17.2%.
But here’s the catch. Orforglipron is still pre-market. These indirect comparisons use different trial populations and protocols. No one has actually run a direct head-to-head trial between orforglipron and tirzepatide yet. So take the comparison with a grain of salt.
Side Effects: Pretty Similar, Honestly
Both drugs cause stomach issues. That’s just the reality of GLP-1 drugs.
A meta-analysis of five RCTs (over 4,400 people) in Endocrinology, Diabetes & Metabolism found that GI side effects were significantly more common at orforglipron doses of 12 mg or higher. The highest dropout rates were at 24 mg (RR: 4.61) and 36 mg (RR: 3.68).
For tirzepatide, a meta-analysis of 14 RCTs (almost 15,000 patients) showed that GI events were higher than placebo at all doses – but the rates were similar to other GLP-1 drugs.
Bottom line on safety: both have the same class-related side effects, mostly GI and manageable. But orforglipron’s oral format might help with long-term adherence for some patients.
The Manufacturing Angle Nobody Talks About
This is where things get really interesting from a supply chain view.
Tirzepatide is a peptide. It’s made using solid-phase peptide synthesis (SPPS). That process is complex, expensive, and hard to scale. Orforglipron is a small molecule. It uses conventional chemical synthesis – much easier to scale, less capital-intensive, and no cold chain required.
So from a manufacturing standpoint, orforglipron represents a real shift. No injections. No refrigeration. Simpler logistics. That’s good for patients, sure – but it’s also good for the entire supply chain.
At Tianming Pharmaceutical, we’ve been developing high-purity intermediates for next-gen metabolic therapies, including oral small-molecule GLP-1 agonists. The move toward oral small molecules for chronic conditions like obesity and diabetes means the industry is going to need a robust, scalable intermediate supply chain. That’s exactly the kind of challenge we’ve built our technical team to solve.
So Which One Is Really “Better”?
If you’re just looking at weight loss at 72 weeks – no contest. Tirzepatide wins. Its dual GIP/GLP-1 mechanism delivers results that no single GLP-1 agonist has matched yet.
But orforglipron isn’t trying to beat tirzepatide at that game. It’s trying to be different: a convenient, oral, small-molecule option that could improve long-term adherence and make treatment more accessible.
Some analysts have even suggested that orforglipron’s real strength might be as a long-term maintenance therapy – because let’s be honest, taking a daily pill is a lot easier for most people than giving yourself a weekly shot.
For someone who really can’t tolerate injections, or who needs a simpler maintenance plan after losing weight with an injectable, orforglipron could end up being exactly what they need.
Final Takeaway
Tirzepatide is the current heavyweight champ – stronger weight loss numbers, more clinical data, already approved. Orforglipron is the challenger with a different strategy: oral, small-molecule, potentially better for long-term adherence, and easier on the supply chain.
Neither is bad. They just solve different problems.
What excites me most, as someone who’s watched the GLP-1 field evolve from the chemistry side, isn’t which molecule wins at 72 weeks. It’s that we now have multiple real approaches – peptide dual agonists, oral small molecules – that can reach patients through different channels. That’s how you tackle a global chronic disease epidemic. Not with one miracle drug. With a toolbox of options.
At Tianming Pharmaceutical, we specialize in high-purity intermediates for next-generation metabolic therapies – both peptide-based and small-molecule GLP-1 agonists. If your pipeline includes oral small-molecule GLP-1 drugs and you’re looking for a reliable intermediate partner, let’s talk. We’re here to help turn promising molecules into dependable products.
Data sources:
Orforglipron ATTAIN-1 trial: Eli Lilly press release (August 2025)
Tirzepatide SURMOUNT-1 trial: Eli Lilly published data
Indirect comparison meta�analysis: Circulation (2024)
GI adverse events meta�analyses: Endocrinology, Diabetes & Metabolism (2024) and Diabetes, Obesity and Metabolism (2023)