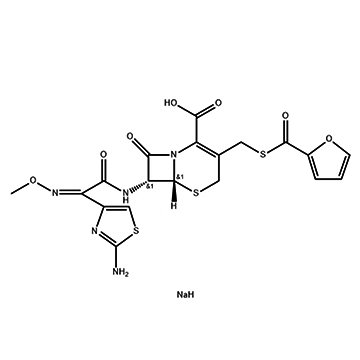
Hydrogenation in Pharmaceutical Intermediates: Risks and Solutions
This article breaks down 4 core risks with authoritative industry data, and shares full-process, FDA & ICH-aligned solutions to reduce accidents, improve yields and ensure compliance.