
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Table of Contents
When it comes to fighting chronic heart failure, Sacubitril Valsartan stands out as a groundbreaking medication. But what exactly makes it so important? Let’s break it down in plain terms.
This medication tackles heart failure in a unique way—it’s like a “two-in-one” therapy. While most heart drugs focus on a single pathway, Sacubitril Valsartan simultaneously targets two key players:
By balancing both, it not only eases symptoms like shortness of breath and fatigue but has been proven to slash hospitalization rates and even extend lives. In fact, clinical trials show it reduces cardiovascular deaths by 20% compared to older therapies.
But here’s the catch: none of this would matter without a reliable supply of high-quality Sacubitril Valsartan. As demand grows globally—driven by an aging population and rising heart disease rates—pharma companies face a pressing question: How do we ensure consistent access to this life-changing ingredient?
The answer starts with understanding its complexity. Producing Sacubitril Valsartan isn’t like baking a cake. It’s a precision-driven process requiring expertise in chemistry, strict quality controls, and a deep grasp of regulatory hurdles.
As demand for Sacubitril Valsartan surges, partnering with a GMP-certified, experienced supplier is critical to securing compliant, high-quality API supplies.
With so many options claiming compliance and quality, how do you separate the best from the rest?
Supplier Evaluation Criteria:
We prioritized suppliers that meet these non-negotiable standards:
At tianming pharmaceutical, we combine: Decades of Expertise in complex API synthesis. Agile Logistics to meet global timelines.Transparent Collaboration from development to delivery.
Take Action Today!

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
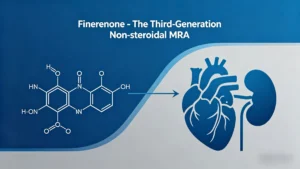
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
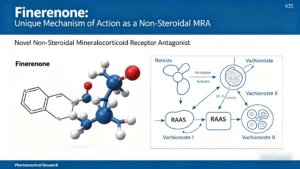
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.