Blog
Follow us for the latest trends, insights and innovations in the pharmaceutical industry.
Certificate of Analysis (COA) for Active Pharmaceutical Ingredients (API): The Authoritative Guide to Quality Assurance
This guide details COA structure (identity, quality, scientific data, compliance), verification checks, common errors, and future-proofing strategies like blockchain tracking. Request a sample COA to ...
Read More →
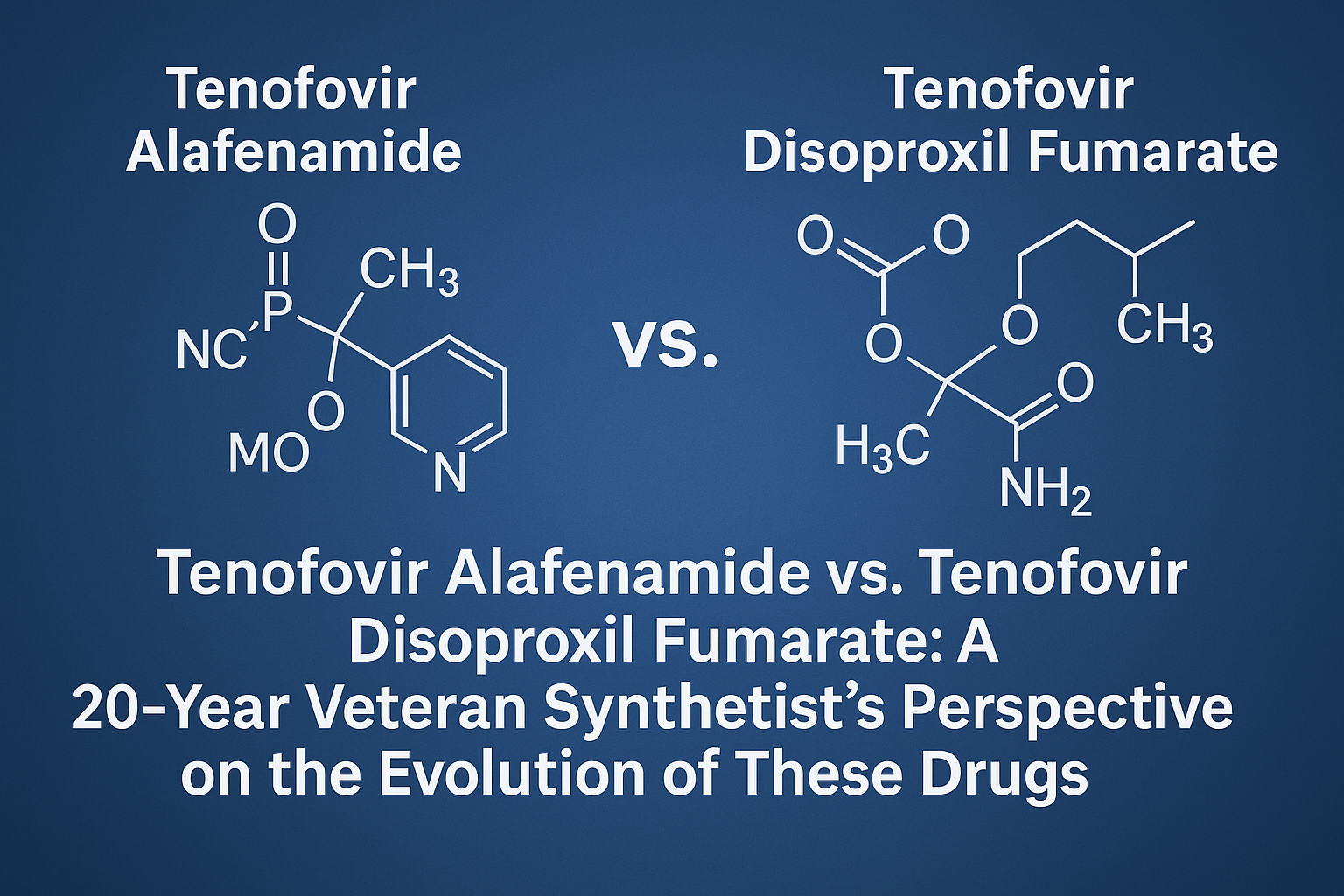
Tenofovir Alafenamide vs. Tenofovir Disoproxil Fumarate: A 20-Year Veteran Synthetist’s Perspective on the Evolution of These Drugs
This expert-authored article compares TAF vs TDF, emphasizing chemical design, safety, & clinical benefits.
Read More →
COA Verification Tools: How to Spot a Fake Certificate of Analysis (and Why It Matters)
Chemical & pharma buyers: Fake COAs risk safety and compliance. Learn manual verification red flags + digital COA verification tools (supplier portals, QR codes, blockchain). ...
Read More →
Pharmaceutical Intermediates Manufacturers: The Indispensable Hidden Champions in the Global Pharmaceutical Supply Chain
This article explores the critical role of pharmaceutical intermediates manufacturers in global drug production, emphasizing GMP compliance, custom synthesis capabilities, and regulatory expertise. It guides ...
Read More →