
Regulatory Differences Between API and Intermediate Manufacturing: A Practical Guide
Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Over the years, I’ve worked my way up in this industry—from sitting in suppliers’ conference rooms undergoing audits to now leading teams to audit others. This role reversal has given me a unique perspective on supplier compliance verification.
Today, I want to discuss this seemingly procedural yet intricately complex topic: how to truly assess a pharmaceutical intermediate supplier’s compliance level.
I recall when I first took charge of supply chain management, I naively believed that as long as a supplier could provide a valid Certificate of Analysis (CoA), everything was essentially in order. That changed when we audited a supplier following standard procedures. Their CoA data was flawless, and their audit documentation was complete.
Yet, during a critical production phase, a batch of their intermediates showed severe impurity exceedances. An urgent investigation revealed their original lab records didn’t match the reported data—the “perfect CoA” was meticulously doctored. That lesson taught me: supplier compliance verification must never stop at paperwork.
Now, whenever I see a polished CoA, my first reaction isn’t delight but caution. Does a comprehensive quality system underpin this document?
My approach is:
Before visiting the site, I dedicate significant time to analyzing the supplier’s documentation system. This is akin to piecing together clues before solving a case:
Once on-site, I typically deviate from the scheduled route to focus on these areas:
I have a routine practice: randomly ask an operator to retrieve a chromatogram and then trace the complete audit trail on the spot. Once, this simple action revealed they used a generic login account, making it impossible to trace the specific operator—a serious data integrity issue.
Review equipment maintenance logs to verify timely and genuine preventive maintenance. Once, beside a reactor, I noticed a pressure gauge calibration sticker expired three months prior—yet they had just completed a critical production batch.
Material management areas most clearly reveal true operational standards. Once, I discovered a batch of poorly labeled materials in a warehouse corner. Upon inquiry, I learned they were non-conforming items awaiting destruction, improperly stored outside designated areas.
I enjoy chatting with frontline operators in production areas, asking questions like, “What do you do if the reaction temperature exceeds limits?” Their responses often reveal operational realities better than any document. Once, a veteran operator detailed an emergency procedure that perfectly matched the SOP—teams like this inspire confidence.
For new suppliers, I now insist on a small-batch trial run. It’s like cohabiting before marriage—it uncovers issues that would otherwise remain hidden.
This trial batch isn’t just about product quality; it also evaluates:
Years of experience have taught me that supplier compliance verification isn’t just a technical task—it’s a matter of integrity. Reliable suppliers withstand the strictest scrutiny, viewing each audit as an opportunity for improvement. Those who evade questions or erect defenses often hide problems in the very corners they keep hidden.
In our industry, every action we take impacts the health of the end patient. Choosing a supplier is fundamentally about selecting a partner who shares our commitment to safeguarding quality. Greater caution and deeper scrutiny are not only responsible to our company but also to the lives we protect.
This is precisely how we hold ourselves accountable at Tianming Pharmaceutical—our doors are always open to client inspections, because we understand that true trust stems from thorough understanding.
If you’re seeking a reliable intermediate partner, we invite you to visit our facilities and speak with our engineers. We’re confident you’ll experience a distinct quality culture.
Partner Email:sunqian0123@gmail.com
WhatsApp: +8617663713557

Regulatory differences between API and intermediate manufacturing under ICH Q7, FDA, EMA, and NMPA. GMP boundaries, DMF filing, starting material justification.
Finerenone drug class: third‑generation non‑steroidal MRA. Compare selectivity, potency, half‑life, and safety vs spironolactone/eplerenone. FDA‑approved for CKD+T2D and HF with LVEF ≥40%.
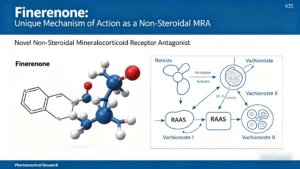
Finerenone mechanism of action: a non‑steroidal mineralocorticoid receptor antagonist with high MR selectivity, balanced cardiorenal distribution, no active metabolites, and lower hyperkalemia risk vs steroidal MRAs.

Leading provider of high-quality APIs and intermediates. Contact us for innovative solutions and expert support.